Background/Objective(s)
The use of mail order pharmacies is increasing as it is a convenient way for people to receive their medications. Medications are being shipped from state to state under varying temperature conditions. The United States Pharmacopeia chapter 1079 suggests practices that pharmacies should follow when mailing medications to help prevent patients from receiving adulterated medications. However, it is unknown how many of these suggested practices are actually being implemented. This study aimed to evaluate the use of these suggested practices across mail order pharmacies in the US. The results of these study will help to support change in the current regulations of mail order pharmacies to increase patient safety as well as increase equality in the rules followed by retail pharmacies as well mail order pharmacies in regards to storage of medications.
Methods
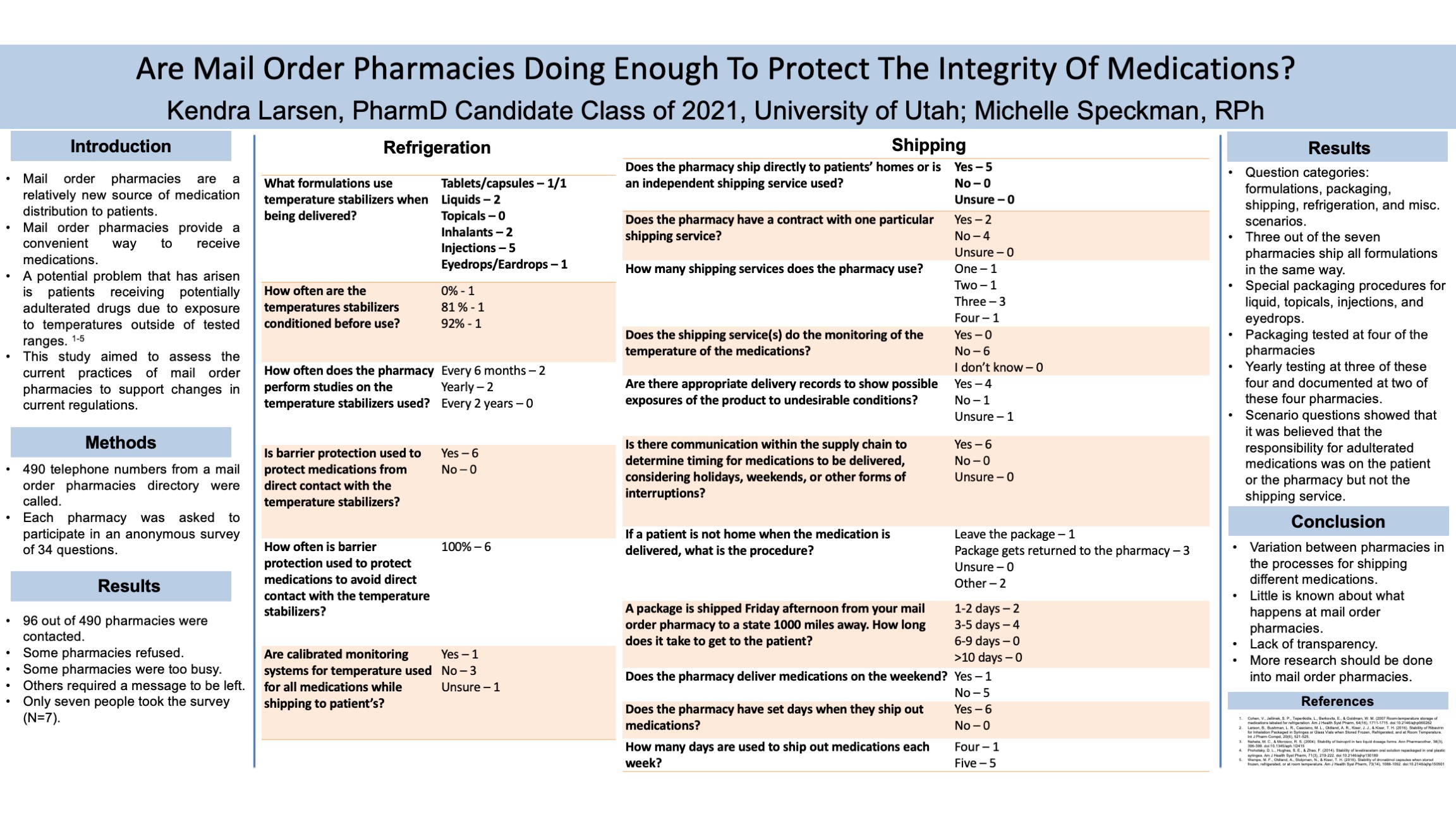
For this evaluative study, a directory of 490 mail order pharmacies was obtained and each pharmacy was called and asked if they would be willing to take a survey about the current practices of their mail order pharmacy. A survey of 32 questions was then sent out to the mail order pharmacies in the US. The questions in the survey asked how often different procedures were completed at the pharmacy such as, how often are continuous temperature trackers used when mailing medications. Qualtrics was used to make the survey and provided the link to send the anonymous survey to these mail order pharmacies. Descriptive statistics will be used to analyze the data to help support the change in regulation of these pharmacies.
Results
Of the 490 mail order pharmacy numbers called, there were seven participants that answered the survey questions. All seven of the participants worked at a mail order pharmacy. Five responses were from a pharmacy manager and two were from pharmacists. The cities and states varied. Three out of the seven responses indicated that all dosage forms are packaged and shipped the same way. The other responses included that liquids, topicals, inhalants, injections, and eyedrops had special packaging procedures. Packaging was tested to ensure that product quality is maintained and there is no damage including environmental or physical damage at four of the seven pharmacies. One pharmacy reported that testing is not done, and two participants were unsure if there was testing completed. Three people indicated that the tests were done yearly and two indicated that these tests are document. Overall, it was shown that there are major inconsistencies between just seven pharmacies. Further research needs to be done to determine if these inconsistencies vary among a great number of pharmacies as well.



Responses
Nice work, Kendra! You put a lot of work into this project. If you were going to give some helpful advice to another student who wanted to do a similar project, what would it be?
The hardest part was finding a list of mail order pharmacies. I think if another student wanted to do something similar, I would try to do more searching for multiple directories of mail order pharmacies. The more people you can get to take a survey the better chance for responses.
This sounds like it was a difficult survey to get responses. I can imagine the pharmacies reluctant to respond if it might not make them look good. Do you have any thoughts about what it would take to get a clear understanding of their shipping practices?
I think it would have to be required by the government or there would have to a lot of pressure from the general public to release information about what is being done in regards to shipping practices.
As mail orders increase, this will become increasingly important issue for patients receiving these drugs.
I agree!!
Kendra, Nice work! I’ll echo Dr. Henchey’s comment — I think you had a big mountain to climb getting mail-order pharmacies to reply, given that the questions are likely to raise issues with their business model. Do you have thoughts on how one might gather this information in a way that wouldn’t put the pharmacies, perhaps, on the defensive and thereby increase the response rate to the survey?
I did my best by stating in the phone calls that the responses would not be traced back to a particular pharmacy or person. I also stated this on the first line of my survey. I am not sure what else could have been done to ease the worry of retribution.
Hi Kendra, This is important work! Do you think there should be a government agency that sets regulations and guidelines for shipping drugs? Based on your study, what should be the highest priority regulations for shipping drugs? Do you think a survey like yours coming from a government entity such as the FDA or state licensing agency might get a better response rate?
Comments are closed.