Purpose
Mail-order pharmacy has evolved to meet the growing prescription needs of Americans. Medications are typically mailed in bubble-padded envelopes without any temperature monitors. Thus, the purpose of this study was to evaluate if the shipping methods used by mail-order pharmacies expose the contents of unrefrigerated packages to potentially unsafe temperatures.
Methods
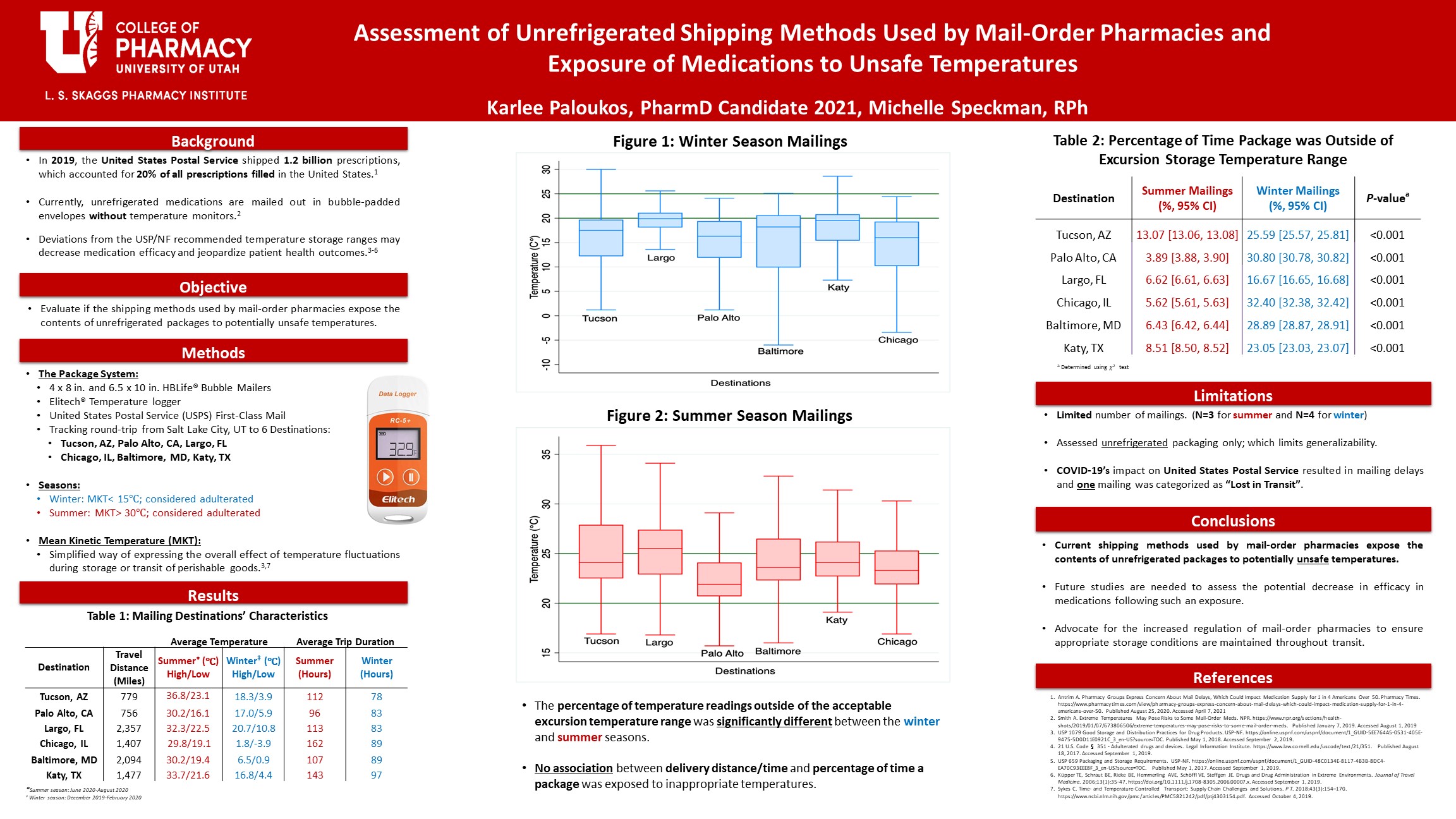
The mean kinetic temperature is used to analyze if a drug product is stored outside of its recommended storage conditions. An Elitech RC-5+ Temperature Data Logger was mailed via USPS first class mail to six destinations: Tucson, AZ; Palo Alto, CA; Largo, FL; Chicago, IL; Baltimore, MD; and Katy, TX. Four mailings occurred during the winter season and three mailings during the summer season. Descriptive statistics were conducted using Microsoft Excel; a Chi-squared test was conducted using Stata software.
Results
All packages fell outside of the acceptable excursion storage temperature range during transit. The percentage of time outside the USP permitted excursion temperature storage range (15°C – 30°C) was 16.67 – 32.40% (p< 0.001) during the winter season and 3.89 – 13.07% (p<0.001) during the summer season (Table 2). There was no association between delivery time and/or travel distance and the percentage of time outside of the acceptable excursion temperature range.
Conclusion
This study shows that the current shipping methods used by mail-order pharmacies expose the contents of unrefrigerated packages to potentially unsafe temperatures. Future studies are needed to assess true causal associations and to further characterize at what temperature and/or length of exposure is associated with decreases in medication efficacy.
Key Words: drug storage, mail-order pharmacy, temperature
Key points:
- The safety and integrity of medications may not be guaranteed as mail-order pharmacy continues to evolve.
- The percentage of time outside the appropriate excursion temperature storage range (15°C -30°C) was 16.67 – 32.40% (p< 0.001) during the winter and 3.89 – 13.07% (p< 0.001) during the summer.
- Future studies are needed to assess true causal associations and to further characterize at what temperature and/or length of exposure is associated with decreases in medication efficacy.



Responses
Awesome job, Karlee! It has been a lot of fun watching this project progress during the various stages. I hope you are well-pleased with the results! What steps should mail order pharmacies take to ensure that temperatures remain within the acceptable range?
Thank you for the question, Dr. Witt! I honestly would have never imagined that this simple project, which I recall diagramming on your office wall whiteboard, would receive such a remarkable response!
I think the first step that mail-order pharmacies should take is to include temperature monitors with all shipments. This would help both the patient and distributor feel more confident that the medication has maintained the acceptable temperature range during transit.
Secondly, mail-order pharmacies should consider evaluating the packaging that medications are shipped in. Much of what we know today regarding the cold chain stems from vaccine storage and less is known with regard to maintaining room temperature storage conditions.
Thank you for your support throughout this endeavor!
Nice work, and it was exciting to see your project get attention at the conference! You suggested that there should be some follow up studies. Do you have any thoughts about what those should look like or how you would design them?
Dr. Henchey, thank you for the question! I would love to conduct a similar study with the 5 most common shipping systems (insulated bubble bag, insulated box liner, EPS molded cooler, insulated envelope, and EPS panel sheet cover). I would like to mail them out again to the different destinations during the different seasons to assess which packaging maintained the USP 1079.2 excursion ranges the best. Furthermore, it would be interesting to mail out a few different formulations (inhaler, tablets, capsules, etc.) and then conduct some form of mass spectrometry to assess if the tablets had truly been degraded by temperature and subsequently lost efficacy.
Wow. Many people I know are receiving mail order, especially since the pandemic isolated them. This study is disconcerting. Thank you.
Thank you for the comment, Jane! It really is a timely subject, especially with Amazon entering the mail-order arena.
Karlee! Your project continues to be so exciting and interesting! I find it curious that the transport times seemed to be greater in the summer than in the winter; I would expect more delays / longer transport times in the winter. Do you have a sense of why that is? Also, is there a sense of how long medications can be out of range before becoming adulterated, or do you think that will be specific for different medications?
Dr. Keefe, thank you for the questions! I agree with your expectation that the winter mailings would yield longer transport times, however; the COVID-19 pandemic had not begun when I mailed out my winter mailing packages. Thus, my study further supports the notion that the USPS was significantly impacted by the pandemic (greater trip durations for all destinations for the summer season), and the media has highlighted various stories of individuals who experienced significant delays when receiving medications by mail, as well as highlighted a few unfortunate situations where some individuals never received certain medications, similar to what occurred with my final winter mailing. 🙁
In regards to your second question, USP 1079.2, which is a new recommendation, as of December 2020, strives to provide recommendations for temperature transit parameters. Essentially, medications can be exposed to a wider excursion temperature range for a short period of time and still be considered “unadulterated”, however; I think this generic recommendation is not fitting for all medications. I would not feel comfortable applying this wide excursion range to medications with narrow therapeutic indexes (levothyroxine, digoxin, etc.) I truly hope in the future that drug manufacturers and/or distributors conduct more thorough stability research on a drug-by-drug basis, especially with Amazon now entering the mail-order realm.
Karlee – Very strong work on this project. What are the two main learning points for you? How would this work be applicable in rural communities? What about during natural disasters? (Katy, TX was particularly affected by the electrical storms this last year)
Dr. Shane-McWhorter, thank you for the questions and for visiting my poster! 🙂
The two main learning points for me were first that sometimes the simplest observations can lead to some very exciting research. I think this point is important for students to understand because we think we need to “cure cancer” to have a successful project, but truly there are things that need improvement in our daily routines that can have a significant impact. Second, one finding that I did not highlight on my poster but I found very interesting was that I only mailed to one mailbox that was “inside”, it was located inside an apartment complex, whereas the other mailboxes were located “outside”. It was surprising to see that the apartment mailbox recorded the coldest temperatures compared to any of the traditional outside mailboxes. Therefore, maybe mailbox type is also a factor that many people may not have considered to play a role?
Rural communities are prime targets for mail-order pharmacies to reach. I think there are many positives to mail-order pharmacy in these areas, including improving adherence, cost-savings, etc. but we need more regulation to ensure that the medications are still efficacious once they reach the patient.
The COVID-19 pandemic impacted my summer mailings (greater trip durations compared to the winter mailings), I think this shows that our current transit mechanisms (USPS in my project) are not adequately prepared to meet the increasing demands for mail-order prescriptions at this time. I would anticipate they would struggle as well to ensure timely deliveries during a natural disaster, therefore, packaging, temperature monitoring, and transit methods need to be further evaluated.
Hi Karlee, Congratulations on the recognition you have received for your work on this project! Your work has raised a number of important questions as indicated by the previous comments in this thread. As a separate issue from monitoring temperatures and improving packaging and/or shipping conditions, do you think it would be worth requiring drug manufacturers to conduct temperature stability studies? This could identify drugs that should not be shipped by mail, and perhaps encourage manufacturers to formulate their drugs to be stable over real-world shipping temperatures.
Dr. Blumenthal, thank you for the question and for visiting my poster! 🙂
I absolutely would advocate for drug manufacturers to conduct more thorough temperature stability studies. Much of my research yielded limited results as far as medication stability during transit. There are a variety of medications (narrow therapeutic index drugs compared to those without a narrow therapeutic index), in addition to different formulations (capsules vs tablets vs inhalers vs creams/ointments). I personally don’t believe that USP 1079.2, which explains the appropriate excursion temperature range during transit, can be viewed as a one-size-fits-all recommendation.
I believe mail-order will continue to evolve as it is appealing to both the consumer and distributors for convenience and cost savings. I anticipate a drone delivering your medications to your front door in our future, especially with Amazon entering the mail-order arena. Thus, I hope my project sparks a conversation and subsequently leads to positive changes in regulation for enhanced patient safety.
Comments are closed.